WO1991012339A2 - Allelic association of the human dopamine (d2) receptor gene in compulsive disorders such as alcoholism - Google Patents
Allelic association of the human dopamine (d2) receptor gene in compulsive disorders such as alcoholism Download PDFInfo
- Publication number
- WO1991012339A2 WO1991012339A2 PCT/US1991/000855 US9100855W WO9112339A2 WO 1991012339 A2 WO1991012339 A2 WO 1991012339A2 US 9100855 W US9100855 W US 9100855W WO 9112339 A2 WO9112339 A2 WO 9112339A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- dna
- allele
- susceptibility
- alcoholism
- human
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/156—Polymorphic or mutational markers
Definitions
- the present invention relates to the first molecular genetic evidence, through the use of RFLP analysis, that an allele in the human dopamine (D 2 ) receptor gene is more significantly associated with human brain tissue from alcoholics than with brain tissue obtained from non- alcoholics.
- the occurrence of this disease-associated polymorphism has a high predictive value in the classification of, at least, one probable subtype of alcoholics.
- the identification of a genetic marker that is closely linked to alcoholism means that the gene's inheritance can be followed, leading to simple tests for diagnosing carriers and future disease victims, and potential gene therapy.
- Alcoholism is a major and devastating health problem with an unknown etiological basis.
- the question of whether environment or heredity is the prime determinant for the development of alcoholism continues to receive extensive attention throughout the world, and has recently involved the Supreme Court of the United States (4) .
- family, twin, and adoption studies (5) are pointing to genetic factors as significant contributors to alcoholism.
- These studies also demonstrate that other forms of mental illness such as schizophrenia and other major psychoses are not found at higher frequencies in families of alcoholics compared with the general population. This would suggest that alcoholism is a primary disease, although many disagree. -3- * -. *•
- Alcoholism currently is viewed as a heterogeneous entity arising from a combination of biopsychosocial factors (6) .
- an extensive literature reveals a wide range of potential physiological (7) and biochemiical (8) markers in the risk for alcoholism.
- family pedigree linkage analysis has implicated chromosomes 4, 6 and 11, but not specific gene markers, in the genetic risk for alcoholism (9).
- Restriction Fragment Length Polymorphism offers a powerful molecular genetic tool for the direct analysis of the human genome to determine elements that provide predisposition to geneti diseases (10) .
- This technique has been used to demonstrate a structural mutation in the gene that codes for an enzyme involved in alcohol metabolism (aldehyde dehydrogenase) which leads to the loss of this enzyme's ability to metabolize acetaldehyde.
- This altered gene is prevalent among Orientals (11) and may a explain the well-known alcohol- flush syndrome as a protective factor in this population.
- no specific gene abnormality has been identified thus far which could regulate alcohol-seeking behavior, or is associated with alcoholism in humans.
- the nigrostriatal is involved in the initiation and execution of movement; the tuberoinfundibular is responsible for the regulation of peptide secretion from the pituitary; and the mesolimbic tract controls emotional stability and affect.
- Mediating these effects of dopamine are two receptor subtypes, D x and D 2 , each of which is coupled to different second messenger systems.
- the D x receptor has been implicated in the sleep disorder, insomnia. Most recently, a D 3 receptor has been found (47) and is also implicated in li bic system function.
- D 2 receptors Important clinically relevant studies on the pharmacology of D 2 receptors indicated that antipsychotic drugs display high affinities for the receptor. Other work suggested that the D 2 receptor is involved in movement disorders, i.e., Parkinson's disease and tardive dyskinesia, tumors of the pituitary, and compulsive disease.
- a cDNA encoding for rat dopamine (D 2 ) receptor has been isolated (15) .
- This receptor has been implicated in the pathophysiology of certain diseases, including drug addiction.
- the same laboratory localized the receptor gene to chromosome 11 (19) .
- Partial sequence analysis revealed that the genomic clone lambda-hD 2 Gl ( ⁇ hD 2 Gl) contains the last coding exon of the D 2 receptor and 16.5 kb of 3-prime flanking sequence.
- the DNA probe ⁇ hD 2 Gl, a 1.6 kb fragment of ⁇ hD 2 Gl, and likely a 30 bit (base pair) fragment of ⁇ hD 2 Gl effectively visualize the human dopamine (D 2 ) receptor gene.
- the present invention concerns a method for diagnosing susceptibility to compulsive disease.
- the method comprises initially obtaining DNA from an individual and determining the presence of a particular human D 2 receptor gene allele. Detection of said allele's presence in the sample is indicative of susceptibility to. compulsive disease, particularly because said allele has been found to be present in a large majority of clinically diagnosed victims of compulsive disease.
- the method is used to indicate a susceptibility to alcoholism, particularly because said allele has been found to be present iii a large majority of clinically diagnosed severe alcoholics.
- the human D 2 receptor gene Al allele is most preferably detected in said DHA.
- the allele is readily detected by digesting DNA with Tag I restriction endonuclease and detecting the presence or absence of human D 2 receptor gene Al allele in the hydrolyzed DNA.
- This method involves separating, preferably according to their size, restriction fragments from the digest. The separated fragments are then probed with labelled lambda-hD 2 Gl or fragment thereof such as a Bam Hl-generated 1.6 kb fragment or a synthetic 30 bit fragment of ⁇ hD 2 Gl (30 base pair oligomer) , for example, to specifically detect the presence of human D 2 receptor gene Al allele.
- the presence of a 6.6 kb fragment in said sample, said 6.6 kb fragment being representative of the human D 2 receptor gene Al allele is indicative of suscep ⁇ tibility to compulsive disease.
- An object of the invention is to provide a safe and reliable method to diagnose alcohol and/or other drug risk at the prenatal and postnatal level.
- the above described method may also be of value in detecting the predisposition toward other compulsive- obsessive behavior patterns including but not limited to overeating or substance abuse such as seen with nicotine, narcotics and other abused drugs.
- the above described method may also be used to detect a susceptibility to attention deficit disorder with hyperactivity (ADDH) in children.
- ADDH attention deficit disorder with hyperactivity
- this method may be correlated to the presence of ADDH and/or to detect susceptibility to alcoholism with greater reliability as well as other genetic diseases such as Tourette Syndrome (48, 49, 50). This genetic disorder has been linked to a severe form of alcoholism possibly *
- Figure 1(A) shows the hybridization pattern of Ta ⁇ I- digested DNA isolated from a heterozygous individual.
- the hybridization probe is the full-length lambda-hd 2 Gl which hybridizes with the 6.6 kb fragment associated with the Al allele plus the 3.7 kb and the 2.9 kb bands associated with the A2 allele.
- the probe also hybridizes with two constant bands, 10.5 and 2.3 kb in length.
- Figure 1(B) shows the hybridization patterns of Tag I-digested DNA isolated from a nonalcoholic (homozygous for the A2 allele) and an alcoholic (heterozygous for the Al and A2 alleles) individual.
- the hybridization probe is a 1.6 kb Bam HI fragment isolated from lambda-hD 2 Gl. Note that the smaller probe does not hybridize to the 2.9 and 2.3 kb Tag I fragments of the human dopamine D 2 receptor gene.
- Lambda-hD 2 Gl is a genomic EMBL 3 phage containing approximately 18 kb of human leukocyte DNA.
- Figure 1(C) schematically shows the production of the 1.5 kb probe for the dopamine D 2 receptor gene obtained from chromosome 11.
- Figure 4 shows saturation curves of [ 3 H]spiperone binding to D2DR in caudates of (A) a nonalcoholic subject homozygous for the A 2 allele (A2A2) and (B) a nonalcoholic subject heterozygous for Al and A2 (A1A2) . Data points are means of duplicate determinations. Details for the binding studies are given in Example 2. Inset: Scatchard analysis of [ 3 H]spiperone binding.
- Figure 6 shows the hybridization patterns of the 1.6 kb probe after doublet separation by subcloning.
- the 1.6 kb fragment (doublet) was ligated by following the procedure given by Sea Plaque GTG agarose and cloned as described in Example 4.
- DNA from antibiotic resistant clones was digested with BamHl and separated by gel electrophoresis as in Figure 5 to identify the presence of the 1.6 kb fragment.
- Other digestions of clone DNA with different endonucleases revealed differences in DNA patterns following separation on gel electrophoresis. In this manner, clones 9 and 16 were selected and grown.
- the 1.6 kb fragment from BamHl-digested DNA from clone 9 and clone 16 was radiolabeled and hybridized with Tag I-digested human genomic DNA using the same procedure as in Figure 1 above.
- the genomic DNA was from two individuals, one homozygous for the A 2 allele, indicated here as A 2 , and one heterozygous, A1A2, indicated as A _
- Figure 7 shows the nucleptide sequence and the hybridization of the 30 Base Pair Oligomer (30 bit fragment of ⁇ hD 2 Gl) to the 1.6 kb probe obtained from Clone 9.
- the 30 base pair oligomer was custom synthesized to correspond to the last 30 base pairs of the cDNA of the 7th exon of the D 2 receptor. This oligomer was radiolabeled and hybridized to the 1.6 kb fragment from clone 9 but not clone 16.
- A2A2 homozygous for the A2 allele lambda-hD 2 Gl ⁇ hD 2 Gl 18 kb probe Used to detect Al allele.
- TE Tris-EDTA Buffer kb Kb Kilobase or Kilobit
- the present invention demonstrates the first allelic association, namely of the dopamine D 2 receptor gene, with alcoholism.
- DNA from matched alcoholic and nonalcoholic brain samples, was digested with restriction endonucleases and probed with the human D 2 receptor gene (lambda-hD 2 Gl) .
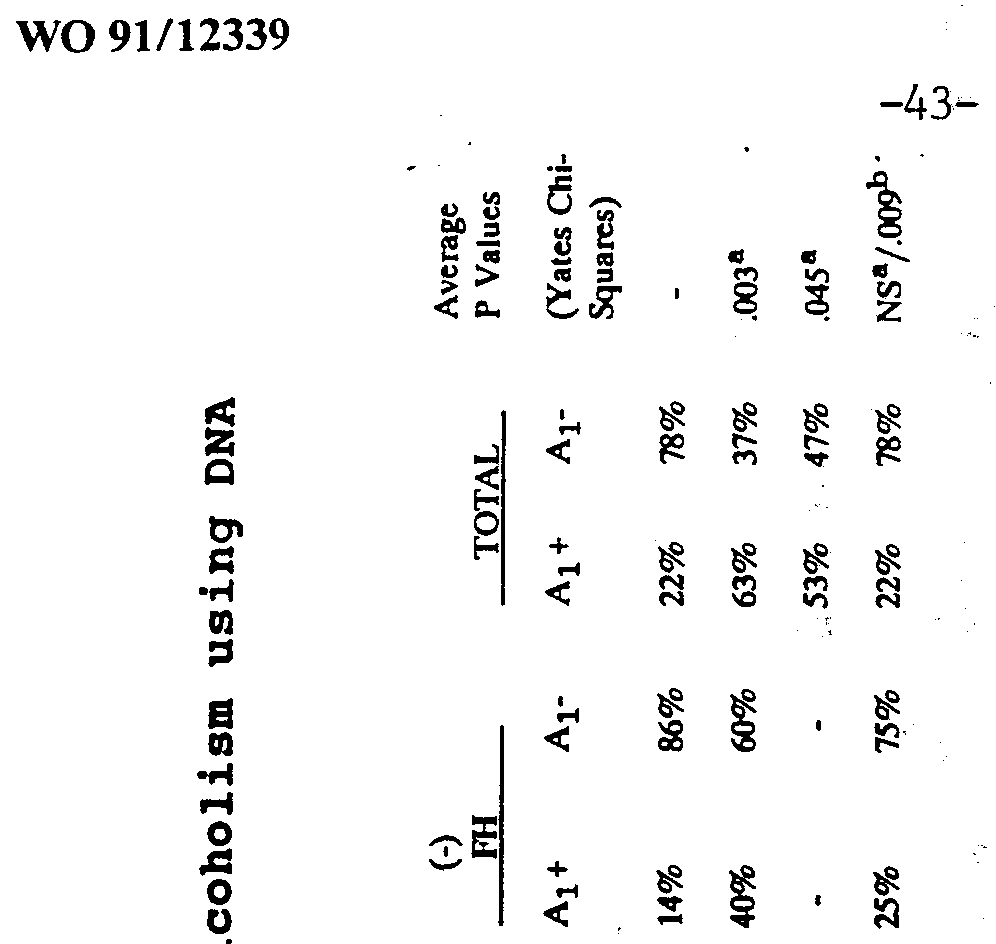
- the presence of the Al (6.6 kb band) allele of the dopamine (D 2 ) receptor (also abbreviated D2DR) gene correctly predicts 77% of alcoholics, and its absence is predictive of 72%.of nonalcoholics.
- the polymorphic pattern of this receptor gene suggests that the abnormality in at least one form of alcoholism is located on the q22-q23 region of chromosome 11 with a co-dominant Mendelian mode of inheritance.
- the allelic association of the dopamine (D 2 ) receptor gene with alcoholism has a high predictive value in the classification of one probable alcoholic subtype. This subtype may represent a virulent form of alcoholism.
- This procedure used to generate the human D 2 receptor gene is as follows: A human genomic library was screened with the rat dopamine D 2 receptor cDNA.
- the human genomic library (Clonetech) in EMBL3 was prepared from normal male leukocyte DNA and screened with a nick- ranslated probe containing portions of the cDNA for the coding region of the rat D 2 receptor.
- One clone, lambda-hD 2 Gl, with an 18 kb insert was identified and characterized. This clone was found to contain the entire 3' coding exon, the polyadenylation signal, and approximately 16.4 kb of noncoding 3' sequence.
- Figure 1 illustrates the polymorphic pattern of the human dopamine (D 2 ) receptor gene.
- Figure 1A depicts the polymorphic allelic pattern for the lambda-hD 2 Gl gene clone.
- Figure IB shows the allelic pattern using a BamHl 1.5 kb subclone which reduced overall background and still was informative as to the presence of alleles Al and A2. However, the smaller probe did not hybridize to the 2.9 and 2.3 kb Tag I fragments of the human dopamine (D 2 ) gene.
- the polymorphic patterns are labelled according to their highly significant allelic association with either alcoholics (Al *
- Table 1 illustrates the polymorphic pattern of the dopamine D 2 receptor gene with DNA obtained from alcoholic and nonalcoholic subjects following three independent hybridizations.
- the Al allele is associated with 24 of 35 (69%) known alcoholics, but it associated with only 7 of 35 (20%) nonalcoholics.
- the absence of the Al allele is associated with 28 out of 35 (80%) of nonalcoholics and with only 11 of 35 (31%) alcoholics.
- Table 2 illustrates the Polymorphic Pattern of the Dopamine D 2 Receptor Gene with DNA obtained from alcoholic and non-alcoholic Caucasians and Blacks following three independent hybridizations.
- N-35 Alcoholic (N-35) N-35
- Nonalcoholic Caucasians N-24
- Alcoholic Caucasians N-ll
- Nonalcoholic Blacks N-ll
- Alcoholic Blacks N-13
- the Al allele is found to be associated with 14 of 22
- Figures 2 and 3 show the samples grouped according to whether or not the Al allele was present. This grouping allowed a classification of samples based on their unique allelic association with alcoholism.
- Figure 2 represents 31 brain samples which possess the Al allele (6.6) . Twenty-four out of 31 DNAs that had the Al allele were from alcoholics. This suggests, that in our sample, the predictive value of this test in correctly identifying alcoholics is 77%.
- Figure 3 represents 39 brain samples which did not possess the Al allele. Since 28 out of 39 samples did not have the Al allele and were from nonalcoholics, this suggests that the predictive value of this test in correctly identifying nonalcoholics is 72%.
- allelic frequencies were 13% and 87% respectively.
- Nuclear DNA was isolated from the matched brain samples as previously described for lambda-hD 2 Gl probe. Twenty micrograms of DNA was digested with one of the four restriction endonucleases. The resulting DNA fragments were separated according to size by electrophoresis in 1% agarose gel, transferred to nitrocellulose membranes, fixed, and hybridized with phosphorus 32 P-labelled probes. Washing of filters and autoradiography were carried out as described previously in this paper.
- a number of probes were employed, including alcohol dehydrogenase (pADH/3) , protein kinase-C (phPKC) , carboxypeptidase-A (CPA) , pro- enkephalin (pHPE9) , tryptophan hydroxylase (TPH479) , tyrosine hydroxylase (BTH , monoamine oxidase B (MAOB) , transferrin (TF) and others (See Table 3) . Evaluation of the data (Table 3) revealed that none of these DNA probes utilizing four restriction endonucleases, (which endonucleases, to date, are responsible for about two- thirds of all known polymorphisms) are associated with alcoholism.
- the cDNA probe for alcohol dehydrogenase an enzyme involved in the metabolism of alcohol, displays a polymorphism using Msp I, but the polymorphism is not linked to alcoholism.
- the cDNA probe for transferrin a protein involved in hemoglobin synthesis, displays polymorphism using Eco RI, but again, this polymorphism is not associated with alcoholism.
- probes used were: protein kinase-C, involved in second messenger coupling mechanisms for neur ⁇ transmitters; carboxypeptidase-A, involved in the metabolism of the opioid peptide enkephalin; pro-enkephalin, the precursor protein for the synthesis of enkephalin; the enzyme tryptophan hydroxylase, involved in the regulation of serotonin synthesis; tyrosine hydroxylase, the rate-limiting enzyme in the synthesis of dopamine; and transferrin, a protein involved in hemoglobin dynamics.
- This latter group of probes as well as others displayed no polymorphism with Tag I, Msp I, Eco RI, and Pst I restriction endonucleases.
- the only probe that showed polymorphism associated with alcoholism was lambda-D 2 Gl.
- Carboxypeptidase A(CPA) no no no no no no - no
- Tyrosine Hydroxylase(BTH 4 ) no no no no no no no no - no
- Serotonin (5HT1A) receptor (GZ1) no no no no no no no - no
- the polymorphism of the dopamine (D 2 ) receptor gene in the brains of alcoholics is due to alcohol-induced alteration in DNA (22) ; hence, the polymorphism observed might be a conseguence of prolonged alcohol consumption by the alcoholic and thus represent a state marker instead of a trait marker. This possibility is unlikely, given the fairly wide prevalence (24%) of the Al allele in the general population. Moreover, the presence of the Al allele and its co-dominant Mendelian inheritance (19) in alcohol-naive children indicate that alcohol per se was not responsible for this genetic variation.
- naive inbred alcohol- preferring rats show a significantly lower dopamine (D 2 ) receptor binding activity than naive alcohol-avoiding rats (23) , suggesting an abnormality in this gene or in its expression.
- D 2 dopamine
- D 2 dopamine
- D2DR human D 2 dopamine receptor
- the allelic association of the human D 2 dopamine receptor (D2DR) gene with the binding characteristics of the D2DR was determined in 66 brains of alcoholic and nonalcoholic subjects.
- the DNA from these samples was treated with the restriction endonuclease Tagl and probed with a 1.5 kb fragment from a BamHl digest of ⁇ hD 2 Gl.
- ⁇ hD 2 Gl contains the entire last exon of the D2DR gene, the polyadenylation signal, and approximately 16.4 kilobases of noncoding 3' sequence of the human D2DR gene.
- the binding characteristics (Kd, binding affinity and Bmax, maximum number of binding sites) of the D2DR were determined in the caudate nucleus from these brains using ( 3 H)spiperone, a dopamine receptor (D2DR) antagonist ligand.
- Log Kd was significantly lower in alcoholic compared to nonalcoholic subjects.
- Bmax was found respectively in A2A2, A1A2, and A1A1 allelic subjects.
- B max the maximum number of D2DRs
- Tissues from 33 alcoholic and 33 nonalcoholic subjects was obtained from the National Neurological Research Bank at the VA Medical Center, Wadsworth, Los Angeles. Frontal grey cortex and caudate nucleus were removed from the brain at autopsy by a neuropathologist and immediately frozen at -70°C until used. The 66 brains analyzed consisted of the 70 that were previously studied in Example I (32) ; 4 caudates were not available. The ages (average ⁇ SEM) of the alcoholics and nonalcoholics, respectively, were 50.4 ⁇ 2.3 years and 53.2 ⁇ 2.6 years. The racial distribution of alcoholics included 21 whites and 12 blacks, and there were 24 white and 9 black nonalcoholics.
- the sex distribution of alcoholics included 30 males and 3 females, and there were 29 male and 4 female nonalcoholics.
- the autolysis times (average ⁇ SEM) of the alcoholics' and nonalcoholics' brain samples were, respectively, 23.0 ⁇ 1.5 hours and 22.6 ⁇ 1.7 hours, alcoholic (Alcohol Dependence and Alcohol Abuse, using DSM-III-R criteria [33]) and nonalcoholic diagnoses were made independently by two trained psychiatrists, through examination of medical and autopsy records, interviews of treatment center personnel and relatives and alcohol consumption data. There was a 100% concordance in diagnosing alcoholic and nonalcoholic subjects between these two assessments. Examination of medical records and/or analysis of body fluids at autopsy did not reveal any of the subjects to have used neuroleptics. The cause of death included: accidents, gun-shot wounds, myocardial infarction, heart failure, cancer, gastrointestinal bleeding, suicide, an pneumonia. Informed consent was obtained from next of kin to carry out the present study.
- the DNA probe as previously used in Example I (32) , was a 1.73 kb band obtained as a doublet from a BamHl digest of a human genomic fragment, ⁇ hD2Gl, provided by 0. Civelli. This fragment contains the last coding (7th) exon of the D2DR gene and part of 16.5 kb of 3• flanking sequence (19,34).
- the 1.5 kb (doublet) probe was labeled by random-priming with [ 32 P]dCTP (18) to a specific activity of 1 x 10 9 cpm/ ⁇ g.
- The. 66 frozen brain cortical samples were coded without reference to their group identity (alcoholic and nonalcoholic) . They were then thawed and processed for high molecular weight genomic DNA and hybridized by established procedures (18) , as previously detailed in Example I (32) .
- D 2 Dopamine Receptor Assay Sixty-six frozen caudate nuclei, of the same brains from which cerebral cortex DNAs were isolated, were also coded without reference to their group identity, and assayed during a one month period for D2DR characteristics. The frozen samples were routinely ground into a fine powder in liquid nitrogen using a mortar and pestle and stored at -70°C from which small amounts of homogeneous powdered tissue could be used for assays at different times.
- a sample of 200-300 mg of the powdered tissue was homogenized in 30 mL of ice-cold buffer (50 mmol/L Tris-HCl, pH 7.4; 120 mmol/L NaCl; 2 mmol/L MgCl 2 ) with a Brinkman Polytron.
- the homogenate was centrifuged at 35,000 x g for 20 minutes.
- the pellet was resuspended in 30 mL of buffer and again centrifuged at 35,000 x g for 20 minutes.
- the final pellet was resuspended in 30 mL of buffer for the binding assay.
- [ 3 H]spiperone (32.4 Ci/mmol; New England Nuclear) in buffer containing 50 mmol/L Tris-HCl, pH 7.4; 120 mmol/L NaCl; 2 mmol/L MgCl 2 .
- S- (-)-sulpiride was added to a final concentration of 10 ⁇ mol/L. Binding was initiated by the addition of membrane preparation (250-350 ⁇ g protein) , and the samples were -27- * • ""
- Al allele consists of both homozygote and heterozygote individuals. These two groups were labeled A1A1 for homozygotes and A1A2 for heterozygotes and were compared with A2A2 homozygote individuals using single factor ANOVA and polynomial tests for trends of B m consult, Log K d and B max /K d .
- [ 3 H]spiperone and Scatchard analysis in caudate tissue (A) non-alcoholic subject with A2A2 allele and (B) nonalcoholic subject with A1A2 allele.
- Table 4A shows the binding characteristics in Al + samples (presence of the 6.6 kb band) and Table 4B depicts the values in Al " samples (absence of the 6.6 kb band). The range of values are consistent with reported results for the binding characteristics of the D2DR in human caudate tissue (42,37,41) .
- Table 5 compares the unadjusted and adjusted K d and B m sanction in caudates of alcoholic and nonalcoholic individuals and that of Al + and Al " allelic subjects.
- Table 5b shows the unadjusted B max of Al + allelic subjects to be significantly lower (P ⁇ .008, one-tailed) than that bt Al " allelic subjects.
- the differences between the alleles for the B m consult measures were parallel between alcoholics and nonalcoholics, thus no factor interaction was detected.
- a test for polynomial trends among the three groups demonstrated a linear relationship (P 0.01, two-tailed) with the highest means for A2A2 followed by the A1A2 samples and with the lowest for the AlAl group.
- Tagl digests of human DNA probed with a clone of a human genomic fragment of the D2DR gene ( ⁇ hD2Gl) , reveal two alleles: Al and A2 (19) .
- the present inventors have shown that digests of DNA, obtained from the cerebral cortex of alcoholics and nonalcoholics, when probed with the 1.6 kb doublet of ⁇ hD2Gl, repealed the Al allele to be associated with alcoholics and its absence to be -34 - * " • ⁇ "
- the caudate nucleus is among brain regions with the highest expression of the D2DR, the guestion raised herein is whether a relationship exists among the binding characteristics of this receptor and the polymorphic pattern of the D2DR gene in caudates of alcoholic and nonalcoholic subjects.
- the polymorphic pattern of this gene and its differential expression of receptors suggests the involvement of the dopaminergic system in conferring susceptibility to at least one subtype of alcoholism.
- EXAMPLE III Following the same procedure described in Example I, the parent clone, lambda-hD 2 Gl, was obtained and digested with BamHl and run on 0.8% agarose (not Sea Plague) and the bands visualized with ethidium bromide staining.
- Figure 5 indicates a doublet in the gel on the right labeled "Doublet 1.73 kb" with DNA molecular weight standards shown in the gel on the left. For egual amounts of different, unrelated fragments, one would expect the smaller fragments to stain with less intensity since fewer nucleotides would be present.
- Doublet 1.73 indicated less staining intensity in the upper, larger kb band ' than in the lower, smaller kb band and since only one band was sometimes observed, it was suspected that the 1.73 kb fragment may consist of two, perhaps related, fragments. Hence, the 1.73 fragment was labeled Doublet 1.73 kb.
- This 1.73 kb fragment was originally estimated by gel electrophoresis to 1.6 kb (personal communication from
- Doublet 1.73 or a 1.73 kb fragment or probe should be considered equivalent to Doublet 1.6 or a 1.6 kb or 1.5kb fragment or probe. Therefore, for discussion purposes in this application, 1.73 kb, 1.6 kb, and 1.5 kb refer to essentially the same fragment, which has now been determined to be 1.6 kb.
- mini-preps were performed and the DNA cut with a series of enzymes (Hinf-1. Msp-1. Tag-1, BamHl, and Hind III) establishing two definite populations containing inserts of the appropriate size (1.6 kb) .
- the 1.6 kb fragments from each clone were labeled with 32 P and used as probes to hybridize onto human genomic DNA cut with Tagl using the same procedure described in Example l.
- Figure 6 shows that the 1.6 kb probe from clone #9 hybridized with human genomic DNA cut with Tagl at 6.6 kb and 3.7 kb, whereas the 1.6 kb probe from clone #16 hybridized with a band at 10.5 kb.
- the 1.6 kb fragment from clone 9 is informative as a probe for the presence of the Al allele in human genomic DNA.
- Another 30 bit oligomer corresponding to the first 30 base pairs of the cDNA coding for the 7th exon of the D2 receptor was tested as a potential probe for the Al allele.
- this oligomer hybridized to the 1.6 kb BamHl fragments (probes) from clones 9 and 16. Therefore, it does not recognize the polymorphism associated with the Al allele.
- the 30 bit probe indicated in Figure 7 was hybridized to the 1.6 kb fragment from clone 9 and not the 6.6 kb fragment from a human genomic DNA, those of skill in the art will recognize that the 30 bit probe will also bind to the 6.6 kb fragment since it comprises a nucleotide sequence which is complementary to the 30 bit probe. Stated another way, since the 1.6 kb probe hybridizes with the Al allele 6.6 kb fragment from human genomic DNA, a subfragment of the 1.6 kb probe would also be expected to hybridize to complementary sequences of the Al allele.
- the 30 bit probe being a complementary subfragment of the 1.6 kb probe, would be recognized by those skilled in the art as capable of hybridizing with a complementary sequence on the 6.6 kb fragment from human genomic DNA thereby being informative as a probe for the Al allele.
- Sequences in the 7th exon of the D2DR gene have been identified that are informative for the Al allele through their ability to hybridize with the 6.6 kb fragment of human genomic DNA. Those sequences are shown in the present invention to be found in a ⁇ hD 2 Gl probe, a 1.6 kb probe of clone 9 and a 30 bit fragment which encodes for the terminal portion of the 7th exon of the D2DR gene. Since the 30 bit fragment seguence is common to both the ⁇ hD 2 Gl probe and the 1.6 kb probe of clone 9, it is likely that analogous seguences within ⁇ hD 2 Gl would also be informative as probes for the Al allele.
- an embodiment of the present invention would utilize a subfragment of the 30 bit fragment comprising about 10 base pairs as a probe.
- subfragments comprising about 15, 20, or 25 nucleotide bases of the 30 bit fragment sequence shown in Figure 7 would be utilized as probes to be informative for the Al allele in human genomic DNA.
- EXAMPLE VI Individuals categorized as non-alcoholic, alcoholic, children of alcoholics (COA) , or drug abuser (DA) were tested for the presence of the Al allele. Subjects were assigned to a particular category after they filled out a standard chemical identification diagnostics form for substance abuse developed by Kenneth Blum, E.P. Noble and associates as well as DSMR-3 criteria assessed by a clinician (DSMR-3 form). The 1.6 kb fragment from clone 9 was used as a hybridization probe for detecting the presence or absence of the Al allele in the subject's DNA isolated from lymphocytes. Genomic DNA from lymphocytes was obtained by the following methodology. ISOLATION OF GENOMIC DNA FROM LYMPHOCYTES
- DNA isolation solution [0.3M sucrose, lOmM Tris (pH 7.5), 5mM MgCl 2 , 1% Triton X-100]. Mix by inversion.
- the alcoholics were found in the brain tissue study of Example I to have 69% A1+ and
- the Al allele may be predictive of attention deficit disorder with hyperactivity (ADDH) .
- ADDH attention deficit disorder with hyperactivity
- coupling of a determination for ADDH with the presence of the Al allele may allow for a more reliable detection of alcoholism susceptibility or may be of benefit in identifying a certain subtype of alcoholism.
- the coupling of other indicators with the presence of the Al allele may offer a significant advance in the detection, prediction or diagnosis of a susceptibility for other compulsive disorders.
- Subjects diagnosed as non-alcoholics include alcohol abuse but not dependence or less severe
- Subjects diagnosed as alcoholic include dependence and eet severe
- Subjects are children of alcoholics have at least one biologic parent clinically diagnosed as alcoholic "Subjects are clinically diagnosed as drug abuse ⁇ and drug dependence where alcohol, if used, is not the drug of choice
- the dopamine (D 2 ) receptor gene polymorphism observed herein may also be associated with predilection to other addictive diseases, such as those relating to nicotine, narcotics or other drugs.
- dopamine acts to reduce stress.
- stress occurs in an individual with a normal number of dopamine receptors
- dopamine is released, all of the receptors are filled, and equilibrium is restored.
- the shortage of dopamine receptors interferes with this process and equilibrium is not restored.
- This person may seek alcohol or other substances or stimuli that release dopamine, in the attempt to find relief and pleasure.
- the desired effects do not come, however, because of the shortage of receptors, and the attempt is repeated, leading to aberrant pleasure-seeking behavior.
- the inventors call this concept the stress-dopamine-genotype hypothesis of craving.
- a polymorphism of the dopamine receptor gene associated with alcoholism and its potential association with other addictive diseases is taught by the present invention.
- the use of the ⁇ hD 2 Gl, 1.6 kb, or 30 bit probes for detecting the Al allele polymorphism of Tag I digests are believed to exemplify one approach to detecting the D2DR gene polymorphism associated with alcoholism.
- the specific approach used in the present application by way of 'example does not preclude alternative approaches to polymorphism detection in this gene.
- the present disclosure teaches a polymorphism for the dopamine receptor gene associated with alcoholism which may be detected using other approaches, in addition to that exemplified in the present disclosure by the Al allele polymorphism in Tag I digests of human genomic DNA.
- Bolos et al. detect a polymorphism by amplifying a 3' noncoding region of the dopamine-. receptor gene seguence with PCR (polymerase chain reaction) and separating the amplified fragments by electrophoresis under nondenaturing conditions.
- This approach is stated to reveal polymorphisms that affect the secondary structure of the single DNA strands which are amplified.
- this invention encompass alternative methods of detecting polymorphisms in the dopamine receptor gene which have been shown by the instant invention to be associated with alcoholism in humans.
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002074519A CA2074519C (en) | 1990-02-07 | 1991-02-07 | Allelic association of the dopamine (d2) receptor gene in compulsive disorders such as alcoholism |
| DE69120526T DE69120526T2 (en) | 1990-02-07 | 1991-02-07 | ALLELIC CONNECTION OF THE HUMAN DOPAMINE (D2) RECEPTOR GENE IN COMPULSION DISORDERS LIKE ALCOHOLISM |
| JP91505069A JPH05508072A (en) | 1990-02-07 | 1991-02-07 | Allelic association of human dopamine (D2) receptor gene in alcoholism-like obsessive-compulsive disorder |
| EP91905230A EP0514490B1 (en) | 1990-02-07 | 1991-02-07 | Allelic association of the human dopamine (d2) receptor gene in compulsive disorders such as alcoholism |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US47705790A | 1990-02-07 | 1990-02-07 | |
| US477,057 | 1990-02-07 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO1991012339A2 true WO1991012339A2 (en) | 1991-08-22 |
| WO1991012339A3 WO1991012339A3 (en) | 1991-10-03 |
Family
ID=23894338
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1991/000855 WO1991012339A2 (en) | 1990-02-07 | 1991-02-07 | Allelic association of the human dopamine (d2) receptor gene in compulsive disorders such as alcoholism |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP0514490B1 (en) |
| JP (1) | JPH05508072A (en) |
| AT (1) | ATE139803T1 (en) |
| CA (1) | CA2074519C (en) |
| DE (1) | DE69120526T2 (en) |
| DK (1) | DK0514490T3 (en) |
| WO (1) | WO1991012339A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1994003602A2 (en) * | 1992-08-10 | 1994-02-17 | STATE OF OREGON, acting by and through THE OREGON STATE BOARD OF HIGHER EDUCATION on behalf of THE OREGON HEALTH SCIENCES DIVISION | A novel human dopamine receptor and its uses |
| US7022816B2 (en) | 1988-11-18 | 2006-04-04 | Oregon Health And Science University | Dopamine receptors and genes |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0771645B1 (en) | 1995-10-31 | 2000-08-23 | Agfa-Gevaert N.V. | On-press development of a lithographic printing plate having an aryldiazosulfonate resin in a photosensitive layer |
| EP0773113B1 (en) | 1995-11-09 | 2000-05-24 | Agfa-Gevaert N.V. | Heat sensitive imaging element and method for making a printing plate therewith |
| EP0773112B1 (en) | 1995-11-09 | 2001-05-30 | Agfa-Gevaert N.V. | Heat sensitive imaging element and method for making a printing plate therewith |
| EP0800928B1 (en) | 1996-04-09 | 1999-07-28 | Agfa-Gevaert N.V. | A heat sensitive imaging element and a method for producing lithographic plates therewith |
| EP0845709B1 (en) | 1996-11-29 | 2006-01-18 | Agfa-Gevaert | A heat sensitive imaging element and a method for producing lithographic plates therewith |
| EP0849091B1 (en) | 1996-12-19 | 2001-03-21 | Agfa-Gevaert N.V. | Heat-sensitive imaging element for making lithographic printing plates comprising polymer particles with a specific particle size |
| EP0849090A3 (en) | 1996-12-19 | 1998-07-01 | Agfa-Gevaert N.V. | Thermosensitive imaging element for the preparation of lithographic printing plates with improved transporting properties |
| DE69901642T3 (en) | 1998-03-14 | 2019-03-21 | Agfa Nv | A process for producing a positive-working printing plate from a thermosensitive image-recording material |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1990005780A1 (en) * | 1988-11-18 | 1990-05-31 | State Of Oregon, Acting By And Through The Oregon State Board Of Higher Education, Acting For And On Behalf Of The Oregon Health Sciences University | Dopamine receptors and genes |
-
1991
- 1991-02-07 AT AT91905230T patent/ATE139803T1/en not_active IP Right Cessation
- 1991-02-07 EP EP91905230A patent/EP0514490B1/en not_active Expired - Lifetime
- 1991-02-07 CA CA002074519A patent/CA2074519C/en not_active Expired - Fee Related
- 1991-02-07 JP JP91505069A patent/JPH05508072A/en active Pending
- 1991-02-07 WO PCT/US1991/000855 patent/WO1991012339A2/en active IP Right Grant
- 1991-02-07 DK DK91905230.8T patent/DK0514490T3/en active
- 1991-02-07 DE DE69120526T patent/DE69120526T2/en not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1990005780A1 (en) * | 1988-11-18 | 1990-05-31 | State Of Oregon, Acting By And Through The Oregon State Board Of Higher Education, Acting For And On Behalf Of The Oregon Health Sciences University | Dopamine receptors and genes |
Non-Patent Citations (4)
| Title |
|---|
| See also references of EP0514490A1 * |
| The American Journal of Human Genetics, volume 45, no. 5, November 1989, University of Chicago Press (Illinois, US), D.K. Grady et al.: "The human dopamine D2 receptor gene is located on chromosome II at q22-q23 and identifies a TaqI RFLP", pages 778-785 * |
| The Journal of the American Medical Association, volume 263, no. 15, 18 April 1990, K. Blum et al.: "Allelic association of human dopamine D2 receptor gene in alcoholism", pages 2055-2060 * |
| The Journal of the American Medical Association, volume 264, no. 24, 26 December 1990, A.M. Bolos et al.: "Population and pedigree studies reveal a lack of association between the dopamine D2 receptor gene and alcoholism", pages 3156-3160 * |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7022816B2 (en) | 1988-11-18 | 2006-04-04 | Oregon Health And Science University | Dopamine receptors and genes |
| US5883226A (en) * | 1990-12-07 | 1999-03-16 | State Of Oregon | Human dopamine receptor and its uses |
| US6203998B1 (en) | 1990-12-07 | 2001-03-20 | Oregon Health Sciences Univ. | Human dopamine receptor and its uses |
| US6437114B1 (en) | 1990-12-07 | 2002-08-20 | Oregon Health & Science University | Human dopamine receptor and uses |
| WO1994003602A2 (en) * | 1992-08-10 | 1994-02-17 | STATE OF OREGON, acting by and through THE OREGON STATE BOARD OF HIGHER EDUCATION on behalf of THE OREGON HEALTH SCIENCES DIVISION | A novel human dopamine receptor and its uses |
| WO1994003602A3 (en) * | 1992-08-10 | 1994-03-31 | Oregon State | A novel human dopamine receptor and its uses |
Also Published As
| Publication number | Publication date |
|---|---|
| WO1991012339A3 (en) | 1991-10-03 |
| EP0514490A1 (en) | 1992-11-25 |
| ATE139803T1 (en) | 1996-07-15 |
| DE69120526D1 (en) | 1996-08-01 |
| CA2074519A1 (en) | 1991-08-08 |
| JPH05508072A (en) | 1993-11-18 |
| DK0514490T3 (en) | 1996-11-04 |
| DE69120526T2 (en) | 1997-02-13 |
| EP0514490B1 (en) | 1996-06-26 |
| CA2074519C (en) | 2003-10-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Blum et al. | Dopamine D2 receptor gene variants: association and linkage studies in impulsive-addictive-compulsive behaviour | |
| RU2195497C2 (en) | Large-scale genotyping of diseases and diagnostic test for cerebellar ataxia of type 6 | |
| AU2014203437B2 (en) | Loci for idiopathic generalized epilepsy, mutations thereof and method using same to assess, diagnose, prognose or treat epilepsy | |
| US5500343A (en) | Allelic association of the human dopamine(D2) receptor gene in compulsive disorders | |
| Mann et al. | The Neurobiology and Genetics of Suicide and Attempted Suicide:: A Focus on the Serotonergic System | |
| Huang et al. | Relationship of psychopathology to the human serotonin1B genotype and receptor binding kinetics in postmortem brain tissue | |
| Sambuughin et al. | Identification and functional characterization of a novel ryanodine receptor mutation causing malignant hyperthermia in North American and South American families | |
| EP0514490B1 (en) | Allelic association of the human dopamine (d2) receptor gene in compulsive disorders such as alcoholism | |
| US5210016A (en) | Allelic association of the human dopamine (D2) receptor gene in compulsive disorders such as alcoholism | |
| Ramesar et al. | Expanded CAG repeats in spinocerebellar ataxia (SCA1) segregate with distinct haplotypes in South African families | |
| Hill | Biological phenotypes associated with individuals at high risk for developing alcohol‐related disorders: Part 1 | |
| Minoretti et al. | A W148R mutation in the human FOXD4 gene segregating with dilated cardiomyopathy, obsessive-compulsive disorder, and suicidality | |
| Goldman | Identifying alcoholism vulnerability alleles | |
| KR101559626B1 (en) | Kit for predicting of therapeutic effect of affective disorder medicine | |
| Nimgaonkar et al. | Schizophrenia and porphobilinogen deaminase gene polymorphisms: an association study | |
| AU2010246325A1 (en) | The SCN2A and SCN3A Loci for Idiopathic Generalized Epilepsy, Mutations thereof and Method using same to Assess, Diagnose, Prognose or treat Epilepsy | |
| Blum et al. | Allelic association of the human dopamine (D 2) receptor gene in compulsive disorders such as alcoholism | |
| WO2002006521A1 (en) | Identification of two principal mutations in ion channels associated with idiopathic generalised epilepsies | |
| WO1996017081A1 (en) | Assessment for treatment with neuroleptic agents | |
| AU667510B2 (en) | DNA encoding a human 5-HT-1F receptor and uses thereof | |
| Maier et al. | Genetics of depression | |
| Masellis | Pharmacogenetic analysis of serotonin receptors and clinical response to clozapine in schizophrenia patients | |
| Blum et al. | 24 The Dopamine D2 Receptor | |
| Maier | Genetics of Mood Disorders: Current Status of Knowledge and Prospects1 | |
| Weight | SYNAPTIC MECHANISMSAND ALCOHOL ACTIONS |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): CA JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IT LU NL SE |
|
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): CA JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): AT BE CH DE DK ES FR GB GR IT LU NL SE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2074519 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1991905230 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1991905230 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1991905230 Country of ref document: EP |