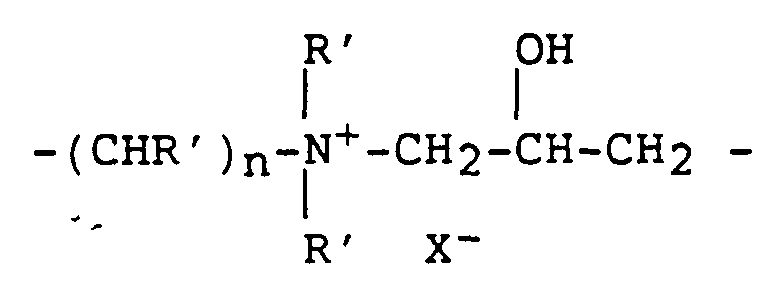WO1994023712A1 - Ionene polymers as anthelmintics in animals - Google Patents
Ionene polymers as anthelmintics in animals Download PDFInfo
- Publication number
- WO1994023712A1 WO1994023712A1 PCT/US1994/003428 US9403428W WO9423712A1 WO 1994023712 A1 WO1994023712 A1 WO 1994023712A1 US 9403428 W US9403428 W US 9403428W WO 9423712 A1 WO9423712 A1 WO 9423712A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- animal
- ionene polymer
- composition
- counter ion
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/14—Quaternary ammonium compounds, e.g. edrophonium, choline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/10—Anthelmintics
Definitions
- This invention relates to the use of ionene polymers as anthelmintics for the treatment of helminth infections in animals .
- helminthiasis The disease or group of diseases described generally as helminthiasis is due to infection of an animal host with parasitic worms known as hel ⁇ rtinths. Helminthiasis is a prevalent and serious economic problem in livestock animals such as swine, sheep, horses, cattle, goats, and poultry. Helminthiasis is also a serious health risk to humans and companion animals such as dogs, cats and other pets.
- Trichostrongylus Certain of these, such as Oesophagostomum, attack primarily the intestinal tract, while others, such as Haemonchus, are more prevalent in the stomach.
- Helminth infections interfere with animal digestion and thus cause anemia, malnutrition, weakness, and weight loss.
- Helminths can also cause severe damage to the walls of the intestinal tract and other tissues and organs and, if left untreated, may result in death of the infected host animal. Accordingly, infected livestock will exhibit poor production performance as manifested by little or no weight gain, metabolic disturbances, sexual cycle abnormalities, and reduced milk production and quality. Parasitic infections detract also from the quality of human and companion animal life. The impact on humans is particularly severe in third world nations. Helminthiasis causes similar symptoms in humans and companion animals to those found in livestock, including nausea, diarrhea, anemia, malnutrition, weight loss, weakness, and, in severe cases, death.
- the invention provides a composition useful in treating helminth infections in animals.
- the invention also provides a method for the treatment of helminth infections in animals.
- the invention prevents the loss of
- the invention improves the health and well-being of companion animals or humans by preventing or
- a method for the treatment of a helminth infection in an animal comprising the step of administering to an animal in recognized need thereof an effective amount of at least one ionene polymer.
- the present invention therefore, relates to a
- composition for the treatment of helminth infections in mammals comprising an effective amount of an ionene polymer and a physiologically acceptable ingredient other than water.
- Ionene polymers or polymeric quaternary ammonium compounds i.e., cationic polymers containing quaternary nitrogens in the polymer backbone (also known as polymeric quats or polyquats), belong to a well-known class of compounds. The biological activity of this class of polymers is also known. See, e.g., A. Rembaum, Biological Activity of Ionene Polymers. Applied Polymer Symposium No. 22, 299-317 (1973).
- Ionene polymers have a variety of uses in aqueous systems such as microbicides, bactericides, algicides, sanitizers, and disinfectants.
- Ionene polymers may be classified according to the repeating unit found in the polymer. This repeating unit results from the reactants used to make the ionene polymer.
- a first type of ionene polymer comprises the repeating unit of formula I :
- R 1 , R 2 , R 3 , and R 4 can be identical or different, and are selected from H, C 1 -C 20 alkyl optionally substituted with at least one hydroxyl group, and benzyl optionally substituted on the benzene moiety with at least one C 1 -C 20 alkyl group.
- R 1 , R 2 , R 3 and R 4 are all methyl or ethyl.
- the group “A” is a divalent radical selected from C 1 -C 10 alkyl, C 2 -C 10 alkenyl, C 2 -C 10 alkynyl, C 1 -C 10 hydroxyalkyl, symmetric or asymmetric di-C 1 -C 10 -alkylether, aryl, aryl-C 1 - C 10 -alkyl, or C 1 -C 10 -alkylaryl-C 1 -C 10 alkyl.
- "A” is C 1 -C 5 alkyl, C 2 -C 5 alkenyl, C 2 -C 5 hydroxyalkyl, or
- the group "B” is a divalent radical selected from C 1 -C 10 alkyl, C 1 -C 10 alkenyl, C 2 -C 10 alkynyl, C 1 -C 10 hydroxyalkyl, aryl, aryl-C 1 -C 10 -alkyl, or C 1 -C 10 -alkylaryl-C 1 -C 10 -alkyl.
- "B” is C 1 -C 5 alkyl, C 2 -C 5 alkeny!, C 2 -C 5
- B is -CH 2 CH 2 -, -CH 2 CH 2 CH 2 -, -CH 2 CH 2 CH 2 CH 2 -, or -CH 2 (CH 2 ) 4 CH 2 -.
- the counter ion, X 2- is a divalent counter ion, two monovalent counter ions, or a fraction of a polyvalent.
- X 2- is two monovalent anions selected from a halide anion and a trihalide anion and more preferably, chloride or bromide.
- Ionene polymers having trihalide counter ions are described in U.S. Patent No. 3,778,476, the disclosure of which is incorporated here by reference.
- I may be prepared by a number of known methods. One method is to react a diamine of the formula R 1 R 2 N-B-NR 1 R 2 with a dihalide of the formula X-A-X. Ionene polymers having this repeating unit and methods for their preparation are
- a second type of ionene polymer comprises the repeating unit of formula II:
- X- is a monovalent counter ion, one half of a divalent counter ion, or a fraction of a polyvalent counter ion sufficient to balance the cationic charge of the repeating unit which forms the ionene polymer.
- X- may be, for example, a halide or trihalide anion, and X- is preferably chloride or bromide.
- a third type of ionene polymer comprises a repeating unit of formula III:
- n and p independently vary from 2 to 12; each R' is
- X 2- is a divalent counter ion, two monovalent counter ions, or a fraction of a polyvalent counter ion sufficient to balance the cationic charge in the group R; and X- is a monovalent counter ion, one half of a divalent counter ion or a fraction of a polyvalent counter ion sufficient to balance the
- R' is hydrogen or C 1 -C 4 alkyl, n is 2-6, and p is 2-6. Most preferably, R' is hydrogen or methyl, n is 3 and p is 2.
- Preferred counter ions for X 2- and X- are the same as those discussed above for formulae I and II.
- the polymers of formula III are derived by known methods from bis-(dialkylaminoalkyl) ureas, which are also known as urea diamines. Ionene polymers of the formula III, methods of their preparation, and their biological activities are described in U.S. Patent No. 4,506,081, the disclosure of which is incorporated here by reference.
- Ionene polymers comprising the repeating units of formulae I, II, and III may also be cross-linked with
- Ionene polymers can be cross-linked either through the quaternary nitrogen atom or through another functional group attached to the polymer backbone or to a side chain.
- Cross-linked ionene polymers, prepared using cross-linking coreactants, are disclosed in U.S. Patent No.
- the cross-linking coreactants listed are ammonia, primary amines, alkylenediamines, polyglycolamines, piperazines, heteroaromatic diamines and aromatic diamines.
- U.S. Patent No. 5,051,124 describes cross-linked ionene polymers resulting from the reaction of dimethylamine, a polyfunctional amine, and epichlorohydrin.
- U.S. Patent No. 5,051,124 also describes methods of inhibiting the growth of microorganisms using such cross-linked ionene polymers.
- the ionene polymers comprising the repeating units of formulae I, II, or III may also be capped, i.e., have a specific amino end group. Capping may be achieved by means known in the art. For example, an excess of either reactant used to make the ionene polymer can be employed to provide a capping group. Alternatively, a calculated quantity of a monofunctional tertiary amine or monofunctional substituted or unsubstituted alkyl halide can be reacted with an ionene polymer to obtain a capped ionene polymer. Ionene polymers can be capped at one or both ends . Capped ionene polymers and their microbicidal properties are described in U.S.
- ionene polymer having a repeating unit of formula I is poly[oxyethylene(dimethyliminio)ethylene- (dimethyliminio)ethylene dichloride.
- R 1 , R 2 , R 3 and R 4 are each methyl, A is -CH 2 CH 2 OCH 2 CH 2 -, B is
- Busan® 77 and WSCP ® are biocides used primarily in aqueous systems, including metalworking fluids, for microorganism control.
- ionene polymer having a repeating unit of formula I is the ionene polymer where R 1 ,
- R 2 , R 3 and R 4 are each methyl, A is -CH 2 CH(OH)CH 2 -, B is - CH 2 CH 2 -, and X 2- is 2Cl-.
- This ionene polymer is a reaction product of N,N,N',N'-tetramethyl-1,2-ethanediamine with
- Busan ® 79 product and WSCP ® II product which are each 60% aqueous solutions of the polymer.
- Preferred ionene polymers having the repeating unit of formula II are those where R 1 and R 2 are each methyl, A is
- the polymer is available from Buckman
- Busan ® 1055 product a 50% aqueous dispersion of the polymer.
- Another preferred ionene polymer having the repeating unit of formula II is obtained as a reaction product of dimethylamine with epichlorohydrin, cross-linked with
- This ionene polymer has a
- X- is Cl- and the ionene polymer is cross-linked with ammonia.
- This ionene polymer has a molecular weight of approximately 100,000-500,000, and is available from Buckman Laboratories, Inc. in a 50% aqueous dispersion sold as the BL ® 1155 product.
- Bubond® 65 are 25% aqueous dispersions of a cross-linked ionene polymer having repeating units of formula II, where R 1 and R 2 are each methyl, A is -CH 2 CH(OH)CH 2 -, X- is Cl-, and the cross-linking agent is monomethylamine.
- This preferred ionene polymer has a molecular weight of approximately
- Preferred ionene polymers having the repeating unit of formula III are those where R is urea diamine and B' is
- the ionene polymer is obtained as a reaction product of N,N'-bis-[1-(3-(dimethylamino)-propyl)] urea and epichlorohydrin, such ionene polymer having an average molecular weight of 2,000-15,000, preferably 3,000-7,000.
- ionene polymers can provide excellent effectiveness against helminth infections caused by nematodes in animals such as livestock and companion animals.
- animal studies described below in the Examples also indicate that ionene polymers can successfully be used in the treatment of helminth infections in humans.
- helminths from the order Strongylida particularly those helminths of the superfamilies Ascaridoidea, Oxyuroidea, Strongyloidea, and Trichostrongyloidea. More particularly, ionene polymers are useful in the treatment of infections caused by the helminths of the families A ⁇ carididae,
- Trichonematidae Trichostrongylidae
- Oesophagostomatinae and even more particularly, helminths of the genera Ascaris, Haemonchus, Oesophagostomum, Syphacia, and Trychostrongylus. Most particularly, ionene polymers are efficacious against the helminths Ascaris suum,
- Oesophagostomum dentatum Oesophagostomum quadrispinulatum
- Syphacia muris Oesophagostomum quadrispinulatum
- Trichostrongylus colubriformis Oesophagostomum dentatum, Oesophagostomum quadrispinulatum, Syphacia muris, and Trichostrongylus colubriformis.
- Ionene polymers also can treat effectively infections by helminths of the order Rhabditida, particularly helminths of the superfamily Rhabditoidea, more particularly helminths of the family Strongyloididae, even more particularly helminths of the genus Strongyloides, and most particularly
- Ionene polymers also can treat effectively infections by helminths of the order Spirurida, particularly helminths of the superfamily Filarioidea, more particularly helminths of the family Onchocercidae, even more particularly helminths of the genus Dirofilaria, and most particularly Dirofilaria immitis (heartworm).
- ionene polymers are effective for treating helminth infections in a variety of host animals, including companion animals and livestock.
- Companion animals include dogs, cats, and horses, as well as less common companion animals such as rodents, birds, and reptiles.
- Livestock animals include monogastrics such as pigs, horses, and poultry, and polygastrics or ruminants such as cattle, sheep, and goats.
- At least one ionene polymer is present in a composition for the treatment of helminth infections in animals in an amount effective to treat helminthiasis, that is, to control the growth or proliferation of nematodes which cause
- the effective amount includes the amount of ionene polymer required to cure or rid an animal of a helminth infection or to decrease the nematode population to an acceptable level not endangering the health of the animal or the desired productivity, in the case of commercial livestock.
- ionene polymers may also be used in a second regimen to
- an effective amount of the ionene polymer may be less than that required to cure or reduce an existing infection, but is an amount effective to prevent helminth infections from occurring.
- the ionene polymer may be administered in one or more doses.
- a second embodiment of the invention therefore, relates to a method for the treatment of a helminth infection in an animal comprising the step of administering to an animal in recognized need thereof an effective amount of at least one ionene polymer.
- This method may be employed in a treatment regimen to cure or rid an animal of a helminth infection or to decrease the nematode population to an acceptable level.
- This method may also be employed as a prophylactic regimen to prevent helminth infections in animals.
- the ionene polymer may be administered in a variety of ways as are known in the art.
- the ionene polymers may be administered orally or, in the case of heartworm, intravenously.
- Oral administration can be in a unit dosage form such as a pellet, tablet, or capsule, or as a liquid drench.
- the drench is normally a solution, suspension, or dispersion of the active ingredient, usually in water, together with a suspending agent such as bentonite and a wetting agent or like excipient. Ethanol is also a suitable solvent.
- the drenches also contain an antifoaming agent.
- the pellets, tablets, or capsules comprise the active ingredient admixed with a physiologically acceptable ingredient vehicle such as starch, talc, magnesium stearate, or di-calcium phosphate.
- physiologically acceptable ingredient is meant one that will not adversely interact with, particularly not react with, the anthelmintic agent and one that may be administered safely to host animals.
- ingredients specifically contemplated for use with this invention are those commonly used in veterinary and
- pharmaceutical practices including, but not limited to, carriers or aids to make the polymers more acceptable to a host animal, flavoring agents, texture agents, odor control agents, adjuvants for other treatments including, but not limited to, vitamins, medicines, medicated or vitamin-enhanced food, weight gain enhancers, other health aids, or any ingredients generally included in food or water for animals.
- pellets, tablets, or capsules containing the desired amount of active compound may be employed.
- These dosage forms are preferably prepared by intimately and uniformly mixing the active ingredient with suitable inert diluents, fillers, disintegrating agents and/or binders.
- Such unit dosage formulations may be varied widely with respect to their total weight and content of the
- antiparasitic agent depending upon factors such as the type of host animal to be treated, the severity and type of infection, and the weight of the host.
- the active compound When the active compound is to be administered via an animal feedstuff, it is preferably intimately dispersed in the feed or used as a top dressing.
- the compound can also be administered in the form of pellets which are added to the finished feed or optionally fed separately.
- the compounds of the invention can also be incorporated into an animal mineral block.
- the anthelmintic compounds of the invention may be administered to animals parenterally, for example, by intraruminal injection, in which event the active ingredient may be dissolved or dispersed in a liquid carrier vehicle.
- the active material may be suitably admixed with a physiologically acceptable vehicle.
- the ionene polymer or polymers When the ionene polymer or a mixture of ionene polymers is administered as a component of the feed or drink of the animals, the ionene polymer or polymers may be dissolved or suspended in drinking water.
- compositions may be provided in which the ionene polymer or polymers are dispersed, preferably intimately, in an inert carrier or diluent.
- a carrier for feed administration is one that is, or may be, an ingredient of the animal ration.
- compositions include feed premixes or supplements in which the active ingredient or ingredients are present in relatively large amounts and which are suitable for direct feeding to the animal or for addition to the feed either directly or after an intermediate dilution or blending step.
- carriers or diluents suitable for such compositions include, for example, distillers' dried grains, corn meal, citrus meal, fermentation residues, ground oyster shells, wheat shorts, molasses solubles, corn cob meal, edible bean mill feed, soya grits, crushed limestone, and the like.
- the active ionene polymers are intimately dispersed throughout the carrier by methods such as grinding, stirring, milling, or tumbling.
- the desired concentration of active compound will vary depending upon the factors previously mentioned as well as upon the particular helminth infection treated and the particular ionene polymer employed.
- individual ionene polymers may be prepared and used. Alternatively, mixtures of two or more individual polymers may be used, as well as mixtures of the polymers and other active compounds not related to the compounds of this invention.
- anthelmintics or methods of treating helminth infections can be combined with the various treatments of helminth infections using ionene polymers according to this invention.
- the other anthelmintics or methods can be
- ionene polymer administered separately or in combination with an ionene polymer to treat or prevent a given helminth infection.
- Use of the same type of treatment regimen for the ionene polymer and other anthelmintic is not required.
- the combination of other anthelmintics or methods with ionene polymer treatment or prophylaxis can have additive or even synergistic efficacy in treating a helminth infection. Examples of such other anthelmintics are fenbendazole, oxfendazole, or the
- Ivermectin® class of avermectins available from Merck & Co.
- mice found to be infected with syphacia muris, were selected for study of five potential anthelmintic compounds.
- Ninety eight rats were divided into eight groups and identified with ionene polymers as follows: Group 1-Busan ® 77, Group 2-Busan ® 1055, Group 3-Busan® 1099, Group 4-Busan ® 1157, Group 5-ASTAT.
- Groups 6 and 7 were used a negative controls and treated with water.
- Group 8 was used as a positive control and treated with piperazine. All animals were dosed by a gavage tube with one treatment per day for four days. Efficacy was determined by standard necropsy and worm counting protocols. Dose rates and
- Trichostrongylus, Cooperia and Oesophagostomum Trichostrongylus, Cooperia and Oesophagostomum.
- Five treatment groups were established: three for experimental compounds, one for a negative control, and one for a positive control. Treatments were administered orally for five consecutive days. Evaluation of efficacy was determined using standard fecal count methods. Efficacy results are given in the Table IV below:
Abstract
Description
Claims
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP52322194A JP3657264B2 (en) | 1993-04-09 | 1994-04-05 | Ionene polymers as animal anthelmintic agents |
| DE69428350T DE69428350T2 (en) | 1993-04-09 | 1994-04-05 | POLYMERS IONES AS ANTHELMINTHICS IN ANIMALS |
| BR9406059A BR9406059A (en) | 1993-04-09 | 1994-04-05 | Pharmaceutical composition and method for treating helminth infection in an animal |
| EP94912342A EP0692964B1 (en) | 1993-04-09 | 1994-04-05 | Ionene polymers as anthelmintics in animals |
| AU64945/94A AU684766B2 (en) | 1993-04-09 | 1994-04-05 | Ionene polymers as anthelmintics in animals |
| CA002159730A CA2159730C (en) | 1993-04-09 | 1994-04-05 | Ionene polymers as anthelmintics in animals |
| AT94912342T ATE205709T1 (en) | 1993-04-09 | 1994-04-05 | POLYMERIC IONENS AS ANTHELMINTICS IN ANIMALS |
| SK1244-95A SK124495A3 (en) | 1993-04-09 | 1994-04-05 | Ionene polymers as anthelmintics in animals |
| NO953995A NO953995L (en) | 1993-04-09 | 1995-10-06 | Ion polymers such as anthelmintics in animals |
| FI954804A FI954804A (en) | 1993-04-09 | 1995-10-09 | Ion polymers as anthelmintics in animals |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/044,597 | 1993-04-09 | ||
| US08/044,597 US5419897A (en) | 1993-04-09 | 1993-04-09 | Ionene polymers as anthelmintics in animals |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1994023712A1 true WO1994023712A1 (en) | 1994-10-27 |
Family
ID=21933240
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1994/003428 WO1994023712A1 (en) | 1993-04-09 | 1994-04-05 | Ionene polymers as anthelmintics in animals |
Country Status (18)
| Country | Link |
|---|---|
| US (1) | US5419897A (en) |
| EP (1) | EP0692964B1 (en) |
| JP (1) | JP3657264B2 (en) |
| CN (1) | CN1078463C (en) |
| AT (1) | ATE205709T1 (en) |
| AU (1) | AU684766B2 (en) |
| BR (1) | BR9406059A (en) |
| CA (1) | CA2159730C (en) |
| CZ (1) | CZ262395A3 (en) |
| DE (1) | DE69428350T2 (en) |
| ES (1) | ES2161762T3 (en) |
| FI (1) | FI954804A (en) |
| NO (1) | NO953995L (en) |
| NZ (1) | NZ263740A (en) |
| PT (1) | PT692964E (en) |
| SG (1) | SG59920A1 (en) |
| SK (1) | SK124495A3 (en) |
| WO (1) | WO1994023712A1 (en) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NZ312601A (en) * | 1995-07-10 | 1999-10-28 | Buckman Labor Inc | A tablet containing an ionene polymer for use in controlling the growth of microorganisms in an aqueous system |
| US5637308A (en) * | 1995-07-10 | 1997-06-10 | Buckman Laboratories International, Inc. | Tabletized ionene polymers |
| US5707534A (en) * | 1995-07-10 | 1998-01-13 | Buckman Laboratories International, Inc. | Use of tabletized ionene polymers in water treatment |
| US5709880A (en) * | 1995-07-10 | 1998-01-20 | Buckman Laboratories International, Inc. | Method of making tabletized ionene polymers |
| US5529700A (en) * | 1995-09-27 | 1996-06-25 | Laporte Water Technologies & Biochem, Inc. | Algicidal or algistatic compositions containing quaternary ammonium polymers |
| WO1997049865A1 (en) * | 1996-06-25 | 1997-12-31 | Buckman Laboratories International, Inc. | Asa sizing emulsions containing low and high molecular weight cationic polymers |
| WO2002056895A2 (en) * | 2001-01-18 | 2002-07-25 | Genzyme Corporation | Ionene polymers and their use in treating mucositis |
| US20040044220A1 (en) * | 2002-08-22 | 2004-03-04 | University Of Florida | Antioxidant and radical scavenging activity of synthetic analogs of desferrithiocin |
| WO2004046109A2 (en) * | 2002-11-19 | 2004-06-03 | Genzyme Corporation | Ionene oligomers and polymers |
| US7803899B2 (en) * | 2005-09-27 | 2010-09-28 | Buckman Laboratories International, Inc. | Methods to reduce organic impurity levels in polymers and products made therefrom |
| AU2008230853A1 (en) * | 2007-03-27 | 2008-10-02 | Buckman Laboratories International, Inc. | Compositions and methods to control the growth of microorganisms in aqueous systems |
| US10836864B2 (en) | 2017-12-12 | 2020-11-17 | International Business Machines Corporation | Chemical compositions with antimicrobial functionality |
| US10667514B2 (en) | 2017-12-12 | 2020-06-02 | International Business Machines Corporation | Antimicrobial ionene compositions with a variety of functional groups |
| US10687528B2 (en) | 2017-12-12 | 2020-06-23 | International Business Machines Corporation | Antimicrobial polymers with enhanced functionalities |
| US10687530B2 (en) | 2017-12-12 | 2020-06-23 | International Business Machines Corporation | Hydrophilic polymers with antimicrobial functionalities |
| US10595527B2 (en) | 2017-12-12 | 2020-03-24 | International Business Machines Corporation | Antimicrobial polymers capable of supramolecular assembly |
| US10743537B2 (en) | 2017-12-12 | 2020-08-18 | International Business Machines Corporation | Monomer compositions with antimicrobial functionality |
| US10653142B2 (en) * | 2017-12-12 | 2020-05-19 | International Business Machines Corporation | Polymers with antimicrobial functionalities |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4025627A (en) * | 1973-12-18 | 1977-05-24 | Millmaster Onyx Corporation | Microbiocidal polymeric quaternary ammonium compounds |
| US4027020A (en) * | 1974-10-29 | 1977-05-31 | Millmaster Onyx Corporation | Randomly terminated capped polymers |
| EP0368593A1 (en) * | 1988-11-04 | 1990-05-16 | Buckman Laboratories International, Inc. | Controlling fungal or bacterial growth in synthetic metalworking fluids |
| US5093078A (en) * | 1989-02-10 | 1992-03-03 | Buckman Laboratories International, Inc. | Novel polymeric quaternary ammonium trihalides and use of polymeric quaternary ammonium trihalides as microbicides, sanitizers and disinfectants |
Family Cites Families (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3562806A (en) * | 1968-09-10 | 1971-02-09 | Eastman Kodak Co | Rumen stable medicament and/or nutrient compositions |
| US4105779A (en) * | 1970-03-10 | 1978-08-08 | Kureha Kagaku Kogyo Kabushiki Kaisha | Anthelmintic process for domestic animals |
| US3898336A (en) * | 1970-05-11 | 1975-08-05 | California Inst Of Techn | Insoluble polymeric quaternary trihalogen salt coated substrates |
| US3778476A (en) * | 1970-05-11 | 1973-12-11 | California Inst Of Techn | Polymeric organic halogen salts |
| US3738945A (en) * | 1972-02-04 | 1973-06-12 | H Panzer | Polyquaternary flocculants |
| USRE28808E (en) * | 1972-02-04 | 1976-05-11 | American Cyanamid Company | Polyquaternary flocculants |
| US3894946A (en) * | 1973-04-02 | 1975-07-15 | American Cyanamid Co | Process for treating industrial wastes |
| US3894947A (en) * | 1973-04-02 | 1975-07-15 | American Cyanamid Co | Process for treating industrial wastes |
| US3930877A (en) * | 1973-08-13 | 1976-01-06 | Nalco Chemical Company | Cationic starch and condensates for making the same |
| US3874870A (en) * | 1973-12-18 | 1975-04-01 | Mill Master Onyx Corp | Microbiocidal polymeric quarternary ammonium compounds |
| US3931319A (en) * | 1974-10-29 | 1976-01-06 | Millmaster Onyx Corporation | Capped polymers |
| US4054542A (en) * | 1975-04-14 | 1977-10-18 | Buckman Laboratories, Inc. | Amine-epichlorohydrin polymeric compositions |
| US4089977A (en) * | 1976-11-24 | 1978-05-16 | Kewanee Industries | Polymeric anti-microbial agent |
| US4147627A (en) * | 1977-02-07 | 1979-04-03 | American Cyanamid Company | Process for scale control using mixtures of polycationic and polyanionic polymers |
| US4164521A (en) * | 1977-02-07 | 1979-08-14 | American Cyanamid Company | Mixtures of polycationic and polyanionic polymers for scale control |
| US4185088A (en) * | 1977-02-17 | 1980-01-22 | Merck & Co., Inc. | Non-adhesive ionene quaternary polymer compositions useful as bile acid sequestrants |
| US4111679A (en) * | 1977-08-17 | 1978-09-05 | Chemed Corporation | Polyquaternary compounds for the control of microbiological growth |
| US4199569A (en) * | 1977-10-03 | 1980-04-22 | Merck & Co., Inc. | Selective hydrogenation products of C-076 compounds and derivatives thereof |
| US4166041A (en) * | 1977-12-15 | 1979-08-28 | American Cyanamid Company | Process for magnesium scale control using mixtures of polycationic and polyanionic polymers |
| US4104161A (en) * | 1978-04-18 | 1978-08-01 | Nalco Chemical Company | Method for treating aqueous wastes containing at least 1% proteinaceous matter |
| AU5957480A (en) * | 1979-07-26 | 1981-01-29 | American Cyanamid Company | Diethyl carbamazine resinate |
| US4352891A (en) * | 1979-09-10 | 1982-10-05 | American Cyanamid Co. | Diethylcarbamazine resinate and styrlpyridinium resinate-diethylcarbamazine resinate edible anthelmintic tablets for companion animals |
| NZ197796A (en) * | 1980-08-04 | 1984-11-09 | Merck & Co Inc | Stabilised aqueous formulations containing ivermectin |
| US4778813A (en) * | 1981-07-07 | 1988-10-18 | Buckman Laboratories International, Inc. | Polymeric quaternary ammonium compounds, their preparation and use |
| US4506081A (en) * | 1982-09-02 | 1985-03-19 | Buckman Laboratories, Inc. | Polymeric quaternary ammonium compounds and their uses |
| US4581058A (en) * | 1982-09-02 | 1986-04-08 | Buckman Laboratories, Inc. | Polymeric quaternary ammonium compounds and their uses |
| US4606773A (en) * | 1984-12-10 | 1986-08-19 | Nalco Chemical Company | Emulsification of alkenyl succinic anhydride sizing agents |
| US4970211A (en) * | 1985-05-20 | 1990-11-13 | Buckman Laboratories International, Inc. | Ionene polymeric compositions, their preparation and use |
| US4758436A (en) * | 1985-05-29 | 1988-07-19 | Merck & Co., Inc. | Drug delivery device which can be retained in the stomach for a controlled period of time |
| FI864812A (en) * | 1985-11-27 | 1987-05-28 | Syntex Inc | FOERFARANDE FOER FRAMSTAELLNING AV AMORFA BENSIMIDAZOLDERIVAT. |
| US4650866A (en) * | 1986-03-06 | 1987-03-17 | Buckman Laboratories, Inc. | Process of preparing 1-methyl-3,5,7-triaza-1-azoniatricyclodecane halides |
| GB8619293D0 (en) * | 1986-08-07 | 1986-09-17 | Munn E A | Anthelmintic agents |
| JPS63297306A (en) * | 1987-05-28 | 1988-12-05 | Katayama Chem Works Co Ltd | Industrial preservative and antifungal agent |
| US4769155A (en) * | 1987-08-19 | 1988-09-06 | Nalco Chemical Company | Turbidity reduction |
| US5128100A (en) * | 1989-10-12 | 1992-07-07 | Buckman Laboratories, Intl., Inc. | Process for inhibiting bacterial adhesion and controlling biological fouling in aqueous systems |
| US5051124A (en) * | 1989-10-24 | 1991-09-24 | Buckman Laboratories International, Inc. | Microbicidal compositions of dimethylamine-epichlorohydrin amines |
| US5145643A (en) * | 1990-01-05 | 1992-09-08 | Allergan, Inc. | Nonoxidative ophthalmic compositions and methods for preserving and using same |
-
1993
- 1993-04-09 US US08/044,597 patent/US5419897A/en not_active Expired - Lifetime
-
1994
- 1994-04-05 CN CN94192382A patent/CN1078463C/en not_active Expired - Fee Related
- 1994-04-05 AU AU64945/94A patent/AU684766B2/en not_active Ceased
- 1994-04-05 AT AT94912342T patent/ATE205709T1/en not_active IP Right Cessation
- 1994-04-05 ES ES94912342T patent/ES2161762T3/en not_active Expired - Lifetime
- 1994-04-05 DE DE69428350T patent/DE69428350T2/en not_active Expired - Fee Related
- 1994-04-05 WO PCT/US1994/003428 patent/WO1994023712A1/en active IP Right Grant
- 1994-04-05 CA CA002159730A patent/CA2159730C/en not_active Expired - Fee Related
- 1994-04-05 SK SK1244-95A patent/SK124495A3/en unknown
- 1994-04-05 SG SG1996000004A patent/SG59920A1/en unknown
- 1994-04-05 CZ CZ952623A patent/CZ262395A3/en unknown
- 1994-04-05 EP EP94912342A patent/EP0692964B1/en not_active Expired - Lifetime
- 1994-04-05 PT PT94912342T patent/PT692964E/en unknown
- 1994-04-05 BR BR9406059A patent/BR9406059A/en not_active Application Discontinuation
- 1994-04-05 NZ NZ263740A patent/NZ263740A/en not_active IP Right Cessation
- 1994-04-05 JP JP52322194A patent/JP3657264B2/en not_active Expired - Fee Related
-
1995
- 1995-10-06 NO NO953995A patent/NO953995L/en not_active Application Discontinuation
- 1995-10-09 FI FI954804A patent/FI954804A/en unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4025627A (en) * | 1973-12-18 | 1977-05-24 | Millmaster Onyx Corporation | Microbiocidal polymeric quaternary ammonium compounds |
| US4027020A (en) * | 1974-10-29 | 1977-05-31 | Millmaster Onyx Corporation | Randomly terminated capped polymers |
| EP0368593A1 (en) * | 1988-11-04 | 1990-05-16 | Buckman Laboratories International, Inc. | Controlling fungal or bacterial growth in synthetic metalworking fluids |
| US5093078A (en) * | 1989-02-10 | 1992-03-03 | Buckman Laboratories International, Inc. | Novel polymeric quaternary ammonium trihalides and use of polymeric quaternary ammonium trihalides as microbicides, sanitizers and disinfectants |
Also Published As
| Publication number | Publication date |
|---|---|
| NO953995D0 (en) | 1995-10-06 |
| DE69428350D1 (en) | 2001-10-25 |
| JPH08511511A (en) | 1996-12-03 |
| FI954804A0 (en) | 1995-10-09 |
| PT692964E (en) | 2002-02-28 |
| CN1078463C (en) | 2002-01-30 |
| SK124495A3 (en) | 1996-06-05 |
| EP0692964B1 (en) | 2001-09-19 |
| DE69428350T2 (en) | 2002-06-13 |
| AU6494594A (en) | 1994-11-08 |
| NO953995L (en) | 1995-12-06 |
| AU684766B2 (en) | 1998-01-08 |
| EP0692964A1 (en) | 1996-01-24 |
| SG59920A1 (en) | 1999-02-22 |
| CA2159730C (en) | 2002-07-16 |
| US5419897A (en) | 1995-05-30 |
| BR9406059A (en) | 1996-01-16 |
| CA2159730A1 (en) | 1994-10-27 |
| JP3657264B2 (en) | 2005-06-08 |
| ATE205709T1 (en) | 2001-10-15 |
| ES2161762T3 (en) | 2001-12-16 |
| CZ262395A3 (en) | 1996-02-14 |
| FI954804A (en) | 1995-11-30 |
| CN1124924A (en) | 1996-06-19 |
| NZ263740A (en) | 1997-08-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0692964B1 (en) | Ionene polymers as anthelmintics in animals | |
| CA1156147A (en) | Treatment of diarrhoea | |
| Wolff et al. | Efficacy of triclabendazole against Fasciola hepatica in sheep and goats | |
| IE59233B1 (en) | Use of biologically active copolymers for the manufacture of a medicament for stimulating the growth of an animal | |
| KR850001495B1 (en) | Anticoccidial combinations | |
| KR101720805B1 (en) | Use of nifurtimox for treating giardiasis | |
| KR100866021B1 (en) | Composition for treatment and prophylaxis of diseases and infections of pigs and poultry | |
| CN102258529A (en) | Compound ivermectin injection and preparation method thereof | |
| Chick et al. | The efficacy of injectable and pour‐on formulations of moxidectin against lice on cattle | |
| US3991209A (en) | Halomethanesulfonamides for eradicating internal parasites | |
| US3150042A (en) | Treating coccidiosis with lincomycin | |
| US3978060A (en) | Method of eradicating internal parasites | |
| NZ207655A (en) | Synergistic veterinary compositions containing an avermectin compound and clorsulon | |
| CZ276893A3 (en) | Pharmaceutical preparation for treating gramm-positive disease even of water animals | |
| CN110898061B (en) | An antibacterial agent suitable for oral administration to animals | |
| EP0107306B1 (en) | Imidazoline derivatives | |
| US20010018417A1 (en) | Virginiamycin mixture | |
| US2702778A (en) | Veterinary tetravalent tin compositions | |
| GB1602745A (en) | Inhibiting growth of mycoplasmas | |
| FI79644B (en) | SYNERGISTIC TILLVAEXTBEFODRANDE FODERTILLSATSAEMNE FOER BOSKAP. | |
| US3968209A (en) | Anthelmintic use of phosphorylated thioureas | |
| US3224939A (en) | Method for treating helminthic infections | |
| KR950009945B1 (en) | "veterinary preparation containing an antibiotic mixture of gentamicin and lincomycin as an additive to drinking water or animal feed and its use in pig breeding | |
| CN114748453A (en) | Bactericidal composition for preventing and treating diseases of aquaculture animals and application thereof | |
| GB2224933A (en) | Synergistic antiparasitic combinations |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 94192382.7 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AT AU BB BG BR BY CA CH CN CZ DE DK ES FI GB GE HU JP KG KP KR KZ LK LU LV MD MG MN MW NL NO NZ PL PT RO RU SD SE SI SK TJ TT UA UZ VN |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 263740 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1994912342 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2159730 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 124495 Country of ref document: SK |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PV1995-2623 Country of ref document: CZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 954804 Country of ref document: FI |
|
| WWP | Wipo information: published in national office |
Ref document number: 1994912342 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: PV1995-2623 Country of ref document: CZ |
|
| WWR | Wipo information: refused in national office |
Ref document number: PV1995-2623 Country of ref document: CZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1994912342 Country of ref document: EP |